Researchers identify brain protein signaling as key to Metformin’s metabolic effects
 Shutterstock
Shutterstock
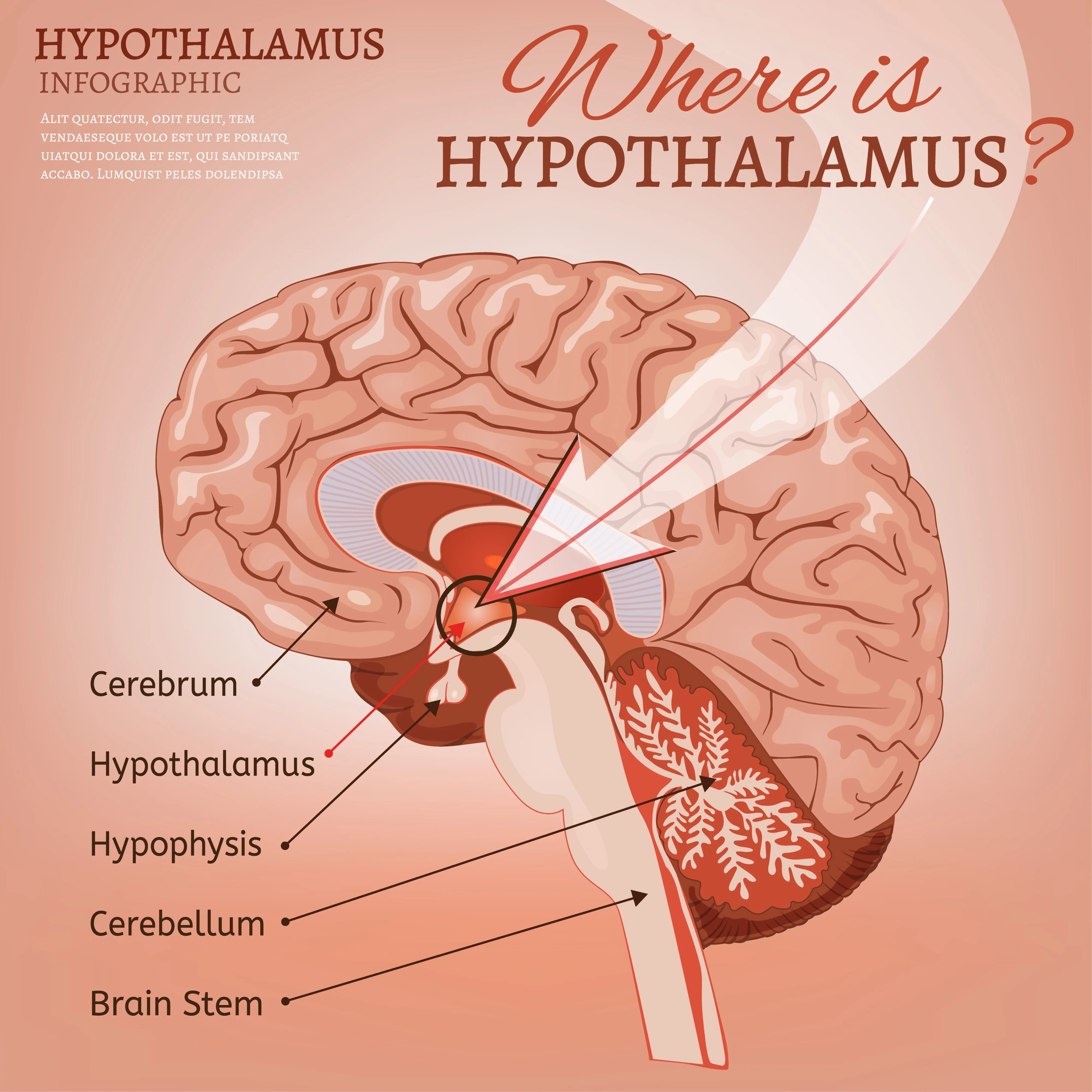
A new study has identified a specific neural pathway that helps explain how Metformin, a frontline treatment for type 2 diabetes used by over 120 million people, regulates blood sugar and body weight. Researchers found that the drug’s effectiveness is significantly tied to the activation of a protein called Rap1 within the hypothalamus, a region of the brain critical for metabolic control.
Rap1 protein signaling facilitates glucose regulation
While Metformin has been prescribed for over 60 years, its primary mechanism was long thought to reside almost exclusively in the liver by suppressing glucose production. However, the recent findings indicate that the drug also targets the brain's "metabolic thermostat."
In laboratory models, when the Rap1 protein was deleted from the hypothalamus, the drug's ability to lower blood glucose and reduce appetite was markedly diminished. This suggests that the communication between the drug and the central nervous system is essential for the full range of its therapeutic benefits.
 Millions of people around the world are prescribed metformin (Scott Olson/Getty Images)
Millions of people around the world are prescribed metformin (Scott Olson/Getty Images)
Hypothalamic interaction clarifies weight loss outcomes
The study provides a clearer biological basis for the weight loss often observed in patients taking the medication. By triggering Rap1 signaling, Metformin appears to enhance the body's sensitivity to leptin, a hormone that signals fullness. According to the research team, this interaction helps explain why the drug is effective at reducing food intake and body weight in addition to managing insulin levels.
 Tests on mice found Metformin had a significant impact on their brains (Getty Stock Photo)
Tests on mice found Metformin had a significant impact on their brains (Getty Stock Photo)
Future implications for metabolic treatment
The discovery of this brain-centered mechanism opens new avenues for developing treatments that could more precisely target metabolic disorders. Understanding that the brain plays a central role in how this common medication functions allows scientists to explore why some patients respond differently to the treatment than others. While the core findings are based on animal models, the conserved nature of the Rap1 pathway in mammals suggests high relevance for human clinical applications and the future of obesity and diabetes research.


Comments (0)
Please login to comment
Sign in to share your thoughts and connect with the community
Loading...