
Navigating the Biochemical Constraints of Vitamin D Co-administration
Vitamin D, specifically Cholecalciferol (D3), operates as a pro-hormone within the endocrine system rather than a simple micronutrient. While the global Health and Wellness Sector has seen a surge in Vitamin D prescriptions to combat deficiencies, the efficacy of these supplements is heavily dependent on the chemical environment of the small intestine.
Recent clinical observations indicate that taking Vitamin D alongside specific minerals or pharmacological agents can lead to competitive inhibition. This occurs when two substances utilize the same transport proteins or metabolic pathways, effectively neutralizing the benefits of one or both components.
The Calcium Paradox and Risks of Hypercalcemia
The most critical interaction involves Calcium supplements. While Vitamin D is required for the body to absorb calcium, an over-saturated environment can trigger a condition known as hypercalcemia. This is characterized by excessive calcium levels in the blood, which can lead to the calcification of soft tissues, including the kidneys and arterial walls.
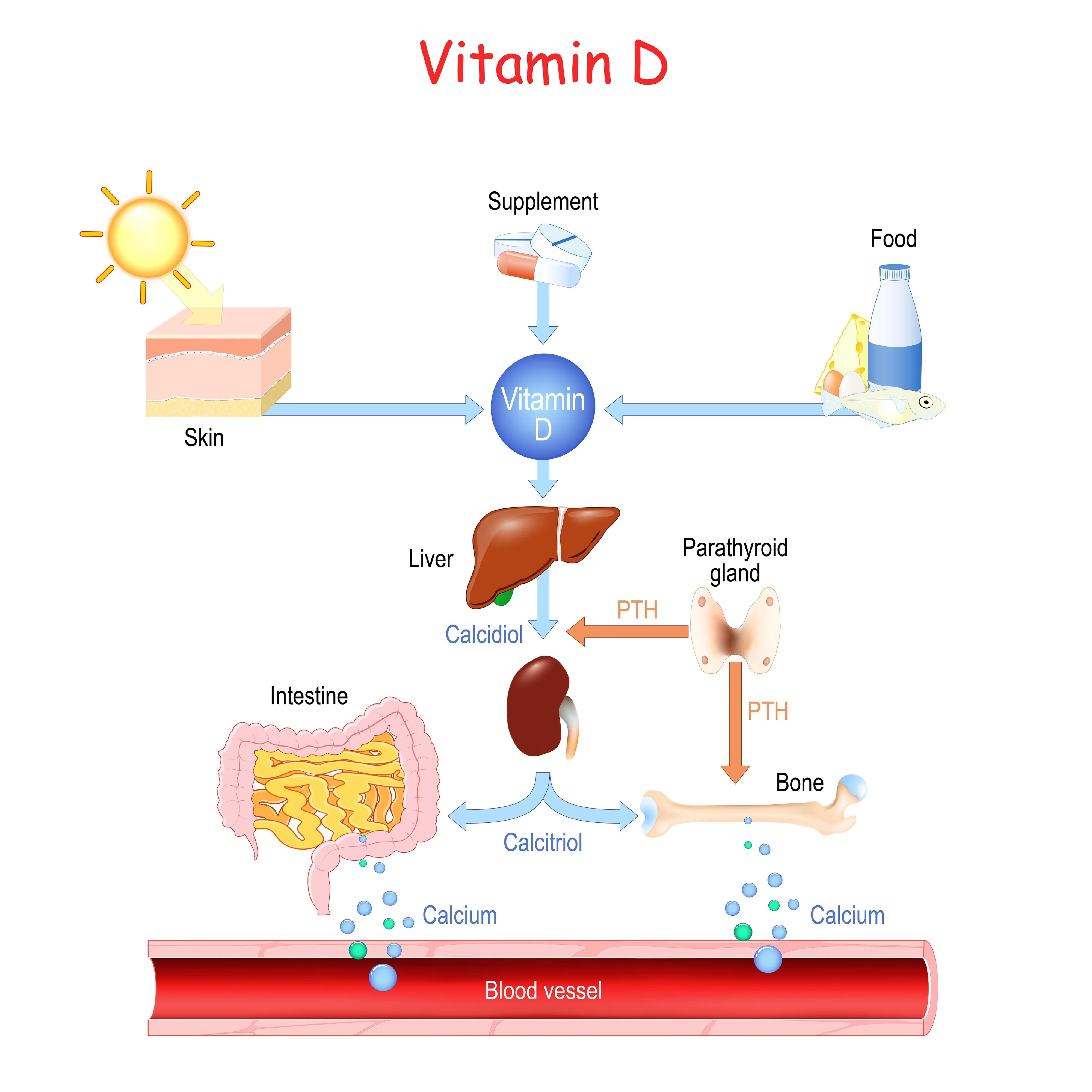
Physicians often recommend a "staggered" approach. Instead of simultaneous ingestion, patients are advised to space these supplements by several hours to allow the Vitamin D Receptor (VDR) to facilitate transport without overwhelming the serum equilibrium.
The Magnesium Sequestration and Metabolic Bottlenecks
In the Nutraceutical Industry, Magnesium and Vitamin D are often marketed together, yet their metabolic relationship is complex. Magnesium is a necessary co-factor for the activation of Vitamin D; however, taking high doses of both simultaneously can create a "competition for absorption" in the intestinal lumen.
If the concentration of Magnesium is too high during the initial digestive phase, it can interfere with the fat-soluble transport mechanism required by Vitamin D. This structural explanation reveals why many users remain deficient in Vitamin D despite high-dosage supplementation they are inadvertently creating a metabolic bottleneck.
Pharmacological Interference: Orlistat and Bile Acid Sequestrators
The interaction between Vitamin D and weight-loss medications like Orlistat (marketed as Alli or Xenical) represents a significant regulatory and safety concern. These drugs function by inhibiting gastric and pancreatic lipases, preventing the absorption of dietary fats.
Because Vitamin D is fat-soluble, the presence of weight-loss stimulants essentially "flushes" the vitamin out of the system before it can be processed. Users in the Pharmaceutical Sector are rarely briefed on this long-term structural shift in nutrient partitioning, which can lead to bone density loss over extended periods of weight-management therapy.
Comparative Interaction and Timing Protocol
| Supplement/Drug | Interaction Type | Biological Mechanism | Recommended Action |
|---|---|---|---|
| Calcium | Synergy/Risk | Increases risk of arterial calcification | Take 4 hours apart |
| Magnesium | Co-factor Competition | Competes for intestinal transport enzymes | Take at different meals |
| Vitamin K | Nutrient Partitioning | Regulates where calcium is deposited | Monitor total intake levels |
| Orlistat | Malabsorption | Inhibits fat-soluble vitamin uptake | Take 2 hours before/after drug |
Systemic Implications for the Global Health Sector
The lack of standardized labeling regarding supplement interactions poses a systemic risk to public health. As the Bio-medical Research community continues to link Vitamin D levels to immune function and mental health, the "more is better" philosophy is being replaced by a focus on "bioavailable timing."
Regulatory bodies such as the Food and Drug Administration (FDA) and the European Food Safety Authority (EFSA) are increasingly under pressure to mandate clearer interaction warnings. Failure to synchronize these nutrients does not just result in "expensive urine" it risks long-term physiological imbalances that can exacerbate the very conditions the supplements were intended to treat.
Future Trajectory of Personalized Micronutrition
The shift toward personalized medicine suggests that the current "one-size-fits-all" approach to Vitamin D is nearing its end. We are moving toward a period of high regulatory uncertainty where the interactions between fat-soluble vitamins and synthetic lipids will require more rigorous clinical validation. As consumers become more aware of the "absorption window," the demand for complex, time-released delivery systems in the supplement market is expected to escalate, potentially disrupting the current low-cost manufacturing models of the nutraceutical industry.


Comments (0)
Please login to comment
Sign in to share your thoughts and connect with the community
Loading...