A systematic review of high-profile Alzheimer’s treatments, including donanemab and lecanemab, has concluded that these drugs do not provide "clinically meaningful" benefits to patients despite their ability to clear brain plaques. The findings suggest a significant disconnect between the biological metrics used in clinical trials and the actual quality of life experienced by those with the disease.
Statistical significance fails the "meaningful change" threshold
The recent review focused on a class of drugs designed to target amyloid-beta plaques, a hallmark of Alzheimer’s disease. While clinical trials for these drugs often report "statistically significant" results, the review argues that these changes are too small to be noticed by patients or their caregivers. To measure this, researchers utilize the Minimum Clinically Important Difference (MCID), a benchmark that identifies the smallest change in a treatment outcome that a patient would identify as important.
The data suggests that while the drugs successfully reduce the volume of amyloid in the brain, the corresponding slowing of cognitive decline falls well below the MCID. In practical terms, a patient on these treatments may show better numbers on a standardized laboratory test compared to a placebo group, but their ability to perform daily tasks or maintain independence remains largely unchanged.
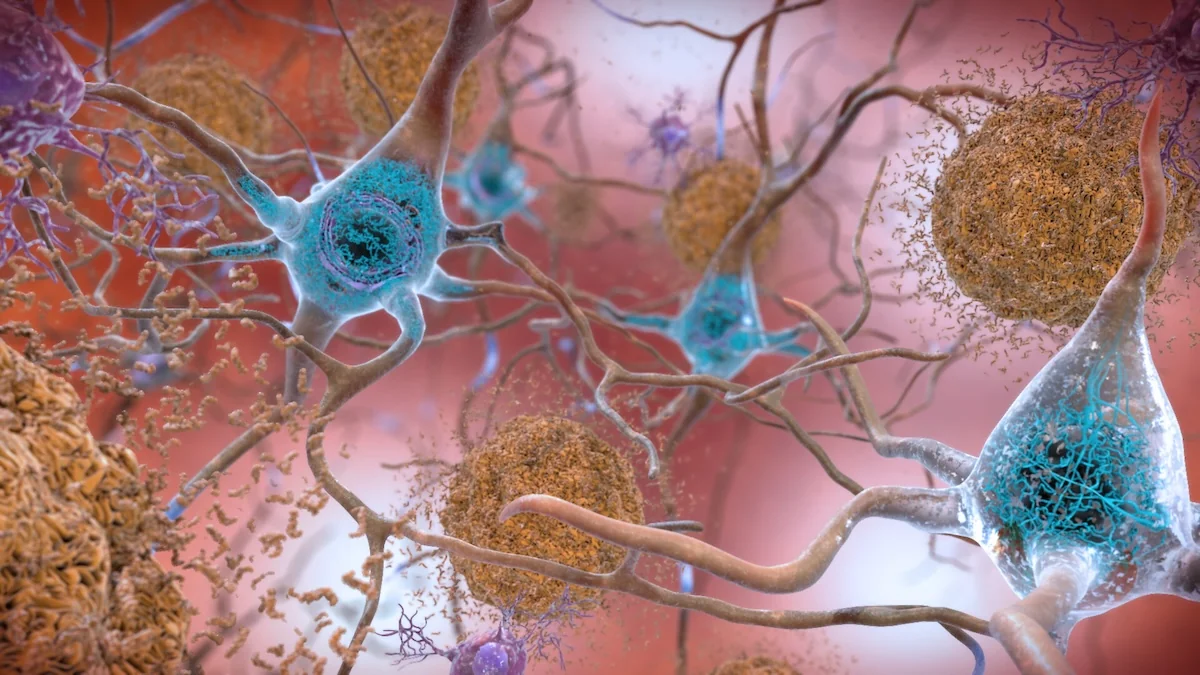 In Alzheimer's, abnormal proteins clump together to form plaques between cells (brown) and inside cells (blue). (NIH/Flickr)
In Alzheimer's, abnormal proteins clump together to form plaques between cells (brown) and inside cells (blue). (NIH/Flickr)
The gap between amyloid clearance and cognitive function
For decades, the "amyloid hypothesis" has driven Alzheimer’s research, based on the premise that removing these protein clusters would halt or reverse the disease. However, this review reinforces a growing skepticism among clinicians regarding the relationship between plaque reduction and clinical health.
The review highlights that the physiological removal of amyloid does not automatically correlate with preserved neurons or improved synaptic function. Practitioners often see a "surrogate endpoint" success where the drug does exactly what it was designed to do at a molecular level without achieving the primary goal of patient care: the stabilization of memory and personality. This discrepancy suggests that amyloid may be a symptom of the disease's progression rather than its sole primary driver.
 The review by the Cochrane organisation looked at drugs that target a plaque called amyloids which builds up in the brains of Alzheimer’s patients. PHOTO: UNSPLASH
The review by the Cochrane organisation looked at drugs that target a plaque called amyloids which builds up in the brains of Alzheimer’s patients. PHOTO: UNSPLASH
Balancing modest slowing against significant safety risks
A critical factor in the review’s conclusion is the weighing of these marginal benefits against the known side effects of anti-amyloid therapies. These drugs are associated with Amyloid-Related Imaging Abnormalities (ARIA), which can manifest as brain swelling or small hemorrhages.
When the clinical benefit is categorized as "not meaningful," the risk-to-reward ratio becomes difficult for many health systems to justify. The review points out that for many patients, the intensive requirements of the treatment including regular intravenous infusions and frequent MRI monitoring to check for brain bleeding may outweigh the negligible slowing of cognitive decline. This assessment places a spotlight on the need for diversifying Alzheimer’s research into alternative pathways, such as inflammation, metabolic dysfunction, or tau protein tangles, rather than continuing to focus exclusively on amyloid.


Comments (0)
Please login to comment
Sign in to share your thoughts and connect with the community
Loading...