Rutgers Study Links Ammonia Surplus to Tumor Acceleration
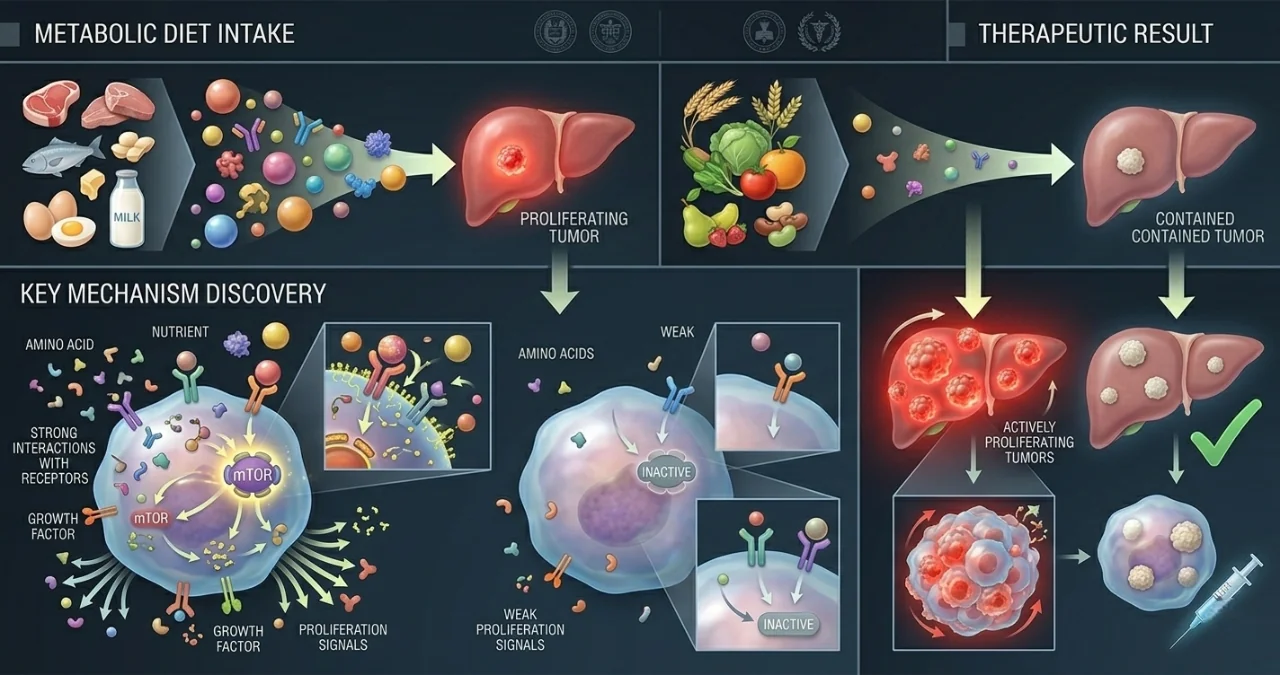
A landmark study led by researchers at Rutgers University has identified a critical metabolic link between dietary protein and the progression of hepatocellular carcinoma (HCC), the most common form of liver cancer. The research, published in early 2026, reveals that when liver function is impaired, the organ loses its ability to convert ammonia—a toxic byproduct of protein digestion—into urea for excretion.
This failure creates a "toxic surplus" of ammonia in the bloodstream. Rather than simply poisoning the host, this ammonia is hijacked by cancer cells to synthesize amino acids and DNA, effectively acting as high-octane fuel for tumor proliferation. This discovery shifts the focus of liver cancer treatment from purely pharmaceutical interventions to precise metabolic management.
The Mechanism of "Ammonia Feeding" in Damaged Livers
In a healthy body, the urea cycle efficiently processes nitrogenous waste. However, in patients with cirrhosis or chronic liver disease, this cycle breaks down. The Rutgers University team found that liver tumors are particularly adept at scavenging this excess nitrogen. By utilizing advanced metabolic tracing, they observed tumor cells "inhaling" ammonia to build the building blocks required for rapid cell division.
In laboratory models involving mice with impaired liver function, the results of dietary intervention were stark. Mice placed on a restricted protein diet showed significantly lower systemic ammonia levels. More importantly, the rate of tumor growth slowed dramatically, and the survival window for the subjects was extended significantly compared to those on a standard high-protein diet.
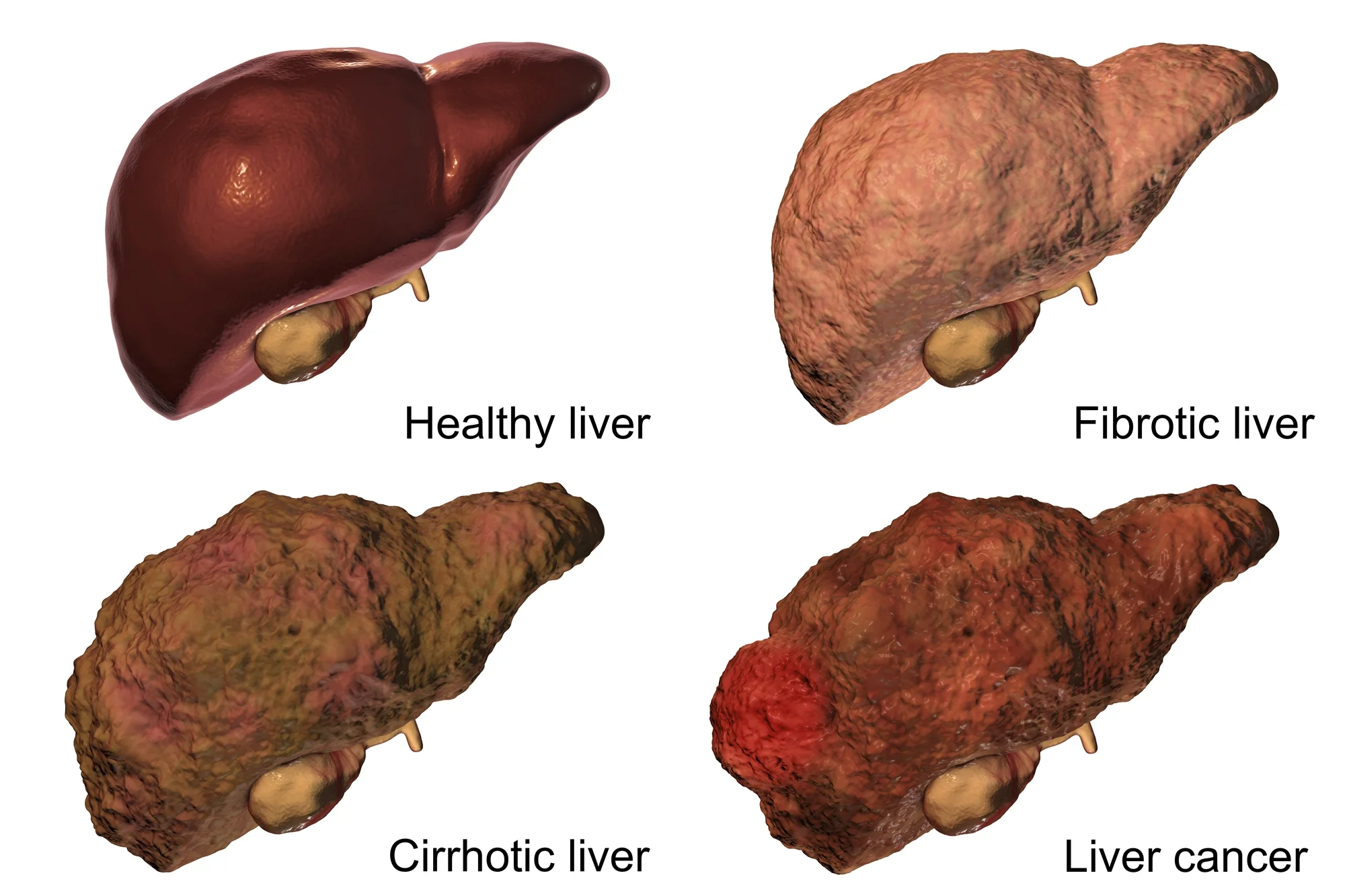 At least 90% of patients with liver cancer are related to liver cirrhosis caused by chronic hepatitis B or C, or excessive-drinking liver disease. In the so-called three-step procedure of “chronic hepatitis →liver cirrhosis→ liver cancer,” the probability of liver cancer generation caused by the DNA or RNA of virus or related protein directly is smaller, while majority is related to the repeated and chronic necrosis-based inflammation in liver. The out-of-control repair later leads to the generation of liver cancer and is related to the timeline and age when suffering the disease
At least 90% of patients with liver cancer are related to liver cirrhosis caused by chronic hepatitis B or C, or excessive-drinking liver disease. In the so-called three-step procedure of “chronic hepatitis →liver cirrhosis→ liver cancer,” the probability of liver cancer generation caused by the DNA or RNA of virus or related protein directly is smaller, while majority is related to the repeated and chronic necrosis-based inflammation in liver. The out-of-control repair later leads to the generation of liver cancer and is related to the timeline and age when suffering the disease
Hidden Implications: The "Protein Paradox" in Cancer Nutrition
Standard oncological advice often emphasizes high protein intake to combat "cachexia" (muscle wasting) and support the immune system during chemotherapy. However, this study introduces a "Protein Paradox" for liver cancer patients. For those with compromised liver function, a standard high-protein diet may inadvertently be "feeding the fire" of the tumor while attempting to save the muscle.
This suggests that the biotech sector and clinical dietetics must move toward "nitrogen-managed" nutrition. The implication is that "protein quality" may matter more than "protein quantity." If researchers can develop protein substitutes or specific amino acid profiles that do not produce excess ammonia during metabolism, they could potentially preserve muscle mass in patients without providing the nitrogenous fuel required by the tumor.
Comparative Impact: High vs. Low Protein in Impaired Livers
| Variable | High Protein Intake | Low Protein Intake |
|---|---|---|
| Ammonia Level | Elevated (Toxic) | Reduced (Controlled) |
| Tumor Fuel Source | High Nitrogen Availability | Nitrogen Starvation |
| Liver Burden | High (Urea Cycle Stress) | Low (Metabolic Relief) |
| Survival Rate | Decreased (in mice) | Significantly Improved (in mice) |
| Metabolic Outcome | Tumor Proliferation | Tumor Deceleration |
Systemic Shift: From Chemotherapy to Metabolic Engineering
The systemic implication of this research is a move toward "metabolic engineering" as a primary pillar of cancer care. By manipulating the patient's internal environment—specifically the nitrogen balance—doctors may be able to make existing treatments like immunotherapy or radiation more effective. A starved tumor is a vulnerable tumor.
This study also highlights a regulatory gap in the healthcare industry. Currently, nutritional interventions are often categorized as "supportive care" rather than "active treatment." The Rutgers findings suggest that for liver cancer, specific dietary protocols should be elevated to the status of a medical prescription, regulated with the same rigor as an oncology drug. This could lead to a surge in specialized medical foods designed for the oncology sector.
What Happens Next: Human Clinical Trials and "Ammonia Stripping"
While the results in mice are conclusive, the transition to human clinical trials is the critical next step. The challenge for researchers will be determining the "Goldilocks Zone" of protein intake—enough to prevent malnutrition but low enough to suppress ammonia.
Parallel to dietary changes, researchers are also looking at "ammonia-stripping" drugs, such as ornithine phenylacetate, which are currently used to treat hepatic encephalopathy. Combining these nitrogen-scavenging drugs with protein-restricted diets could create a dual-action therapy that aggressively targets the tumor's metabolic supply lines. As we move into late 2026, the first human pilot programs are expected to begin in the United States, potentially redefining the standard of care for thousands of patients with liver-impaired cancer.


Comments (0)
Please login to comment
Sign in to share your thoughts and connect with the community
Loading...