Why Tardigrades Survive Without Water: The CAHS Mechanism


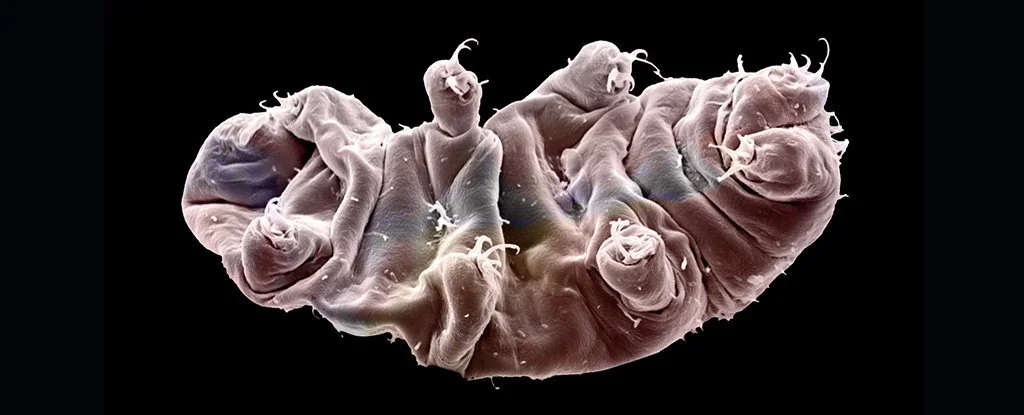
Tardigrades, or "water bears," maintain a legendary status in biology for surviving the vacuum of space and decades of total desiccation. Recent research from the University of Tokyo has pinpointed a unique family of proteins, known as Cytoplasmic Abundant Heat Soluble (CAHS) proteins, as the primary mechanism preventing these organisms from physically shattering when they lose their internal water.
The mechanical role of CAHS proteins in cellular stabilization
Most life forms face a physical crisis during dehydration: as water leaves the cell, internal pressure drops and the cell membrane collapses inward. This mechanical stress typically leads to irreparable structural damage and cell death. However, researchers led by Biologist Takekazu Kunieda and Akihiro Tanaka identified that eutardigrades utilize 336 specific proteins to combat this process.
Among these, CAHS proteins are the most critical. When a tardigrade begins to dry out, these proteins undergo a phase transition, forming a network of gel-like filaments. These filaments act as an internal scaffold, increasing the stiffness of the cytoplasm to resist the external pressure of a shrinking environment. This "bioglass" state effectively holds the cell's internal components in a suspended, rigid architecture until water returns.
Preventing lethal shrinkage through induced stiffness
The University of Tokyo team validated this mechanism by inserting CAHS genes into human and insect cells. Under induced dehydration stress, the modified cells demonstrated a significant increase in survival compared to untreated cells. The CAHS proteins reacted to the loss of water by creating a structural "cage" that prevented the cell membrane from folding or rupturing.
 Preventing lethal shrinkage through induced stiffness
Preventing lethal shrinkage through induced stiffness
From a practitioner’s perspective, this finding shifts the understanding of tardigrade resilience from a purely chemical protection (like antioxidant production) to a mechanical one. The protein doesn't just protect the cell's chemistry; it reinforces its physical integrity. Interestingly, the research showed that this protein also offers protection against osmotic stress caused by excess water, suggesting the CAHS network is a versatile stabilizer for varying pressure environments.
The reversibility of the "bioglass" state
A key limitation in applying this discovery to human medicine—such as organ preservation or "dry" vaccine storage—is the reversibility of the process. In tardigrades, the CAHS filaments dissolve almost instantly when rehydrated, allowing the cell to resume normal metabolic activity without scarring or permanent rigidity.
While the researchers successfully demonstrated increased cell stiffness in human cultures, the full cycle of total desiccation and successful "reanimation" in complex human tissues remains a significant hurdle. The current evidence proves that CAHS proteins can stabilize the cell, but the metabolic "restart" button that tardigrades hit upon rehydration involves a much broader suite of biological signals that are not yet fully understood or replicated in non-tardigrade cells.

Comments (0)
Please login to comment
Sign in to share your thoughts and connect with the community
Loading...